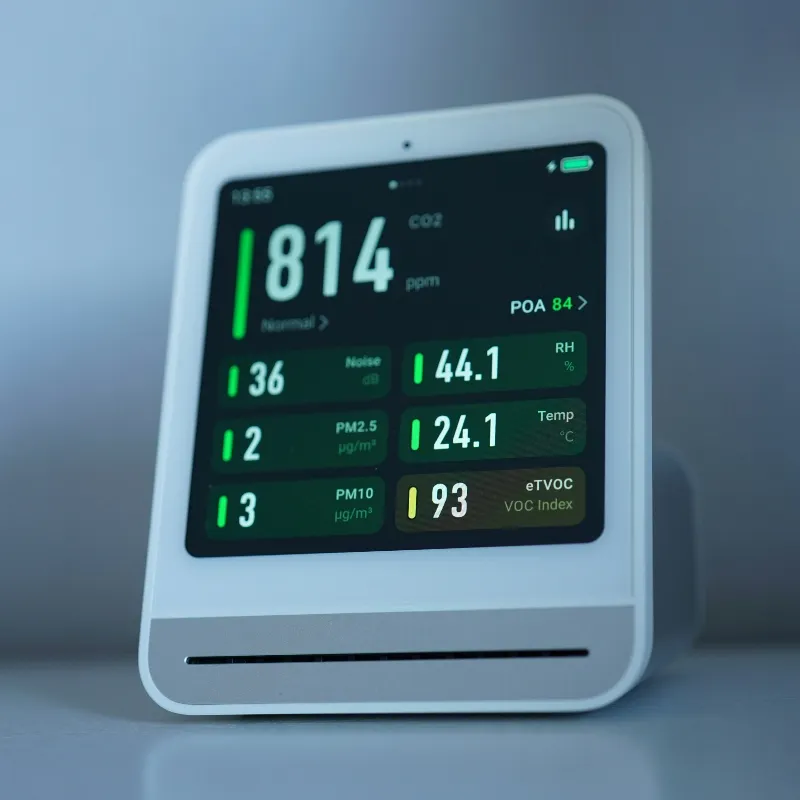
Ensuring Accurate and Dependable Medical Systems
Medical devices must operate reliably under all conditions while protecting patients and users. We support testing for therapeutic devices, diagnostic equipment, and technical medical aids.
From prototype validation to production release, we provide standards-compliant, transparent, and technically precise testing.
Reliable testing. Regulatory confidence. Patient safety.